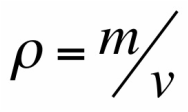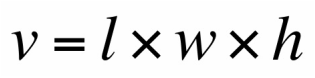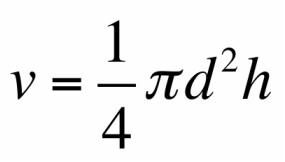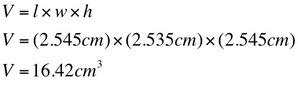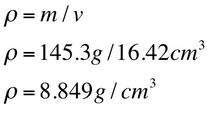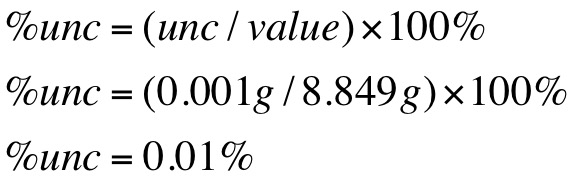Density Lab
Author: Jacob Hoffner
Lab Partners: Andrea Schaefer and Connor Frey
Date: September 2015
Lab Partners: Andrea Schaefer and Connor Frey
Date: September 2015
Purpose
Using various measurements, we will be able to determine the density of several objects.
Theory
Experimental Technique
|
We will determine the density of two rectangular prisms as well as a cylinder. First, we will use a vernier caliper to find the dimensions, and ultimately the volume, of the three objects. Second, we will use a digital balance to find the mass of the three object. We can therefore calculate the densities with the data found. Lastly, we will compare our found values with the known densities of the objects by using percent error.
|
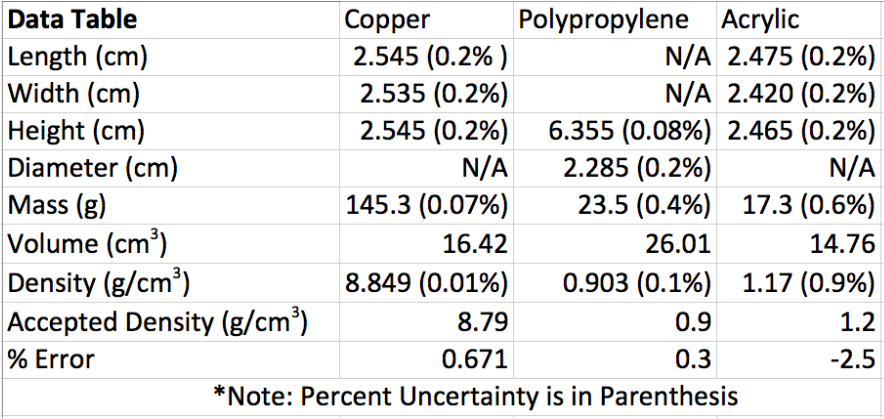
Data
Here are the three objects being measured: Copper Cube (Top Left), Polypropylene Cylinder (Top Right), and an Acrylic Cube (Bottom).
Analysis
Sample Calculations are shown below of density and percent uncertainty.
Conclusion
The main goal of the lab was to determine the density of several objects. My density results were very close to the accepted value of the materials the objects were made out of. The densities I found for the copper rectangular prism and the polypropylene cylinder were just slightly over the accepted value by a percent error of 0.671% and 0.3%, respectively. However, the acrylic rectangular prism was just under the accepted value, receiving a percent error of -2.5%. Errors associated with the measurements made include the possible misjudgment of a dimensional measurement of an object using the vernier caliper to the 0.005 of a centimeter from an observational error. Plus, there is a hole drilled in the polypropylene cylinder, which throws off its mass when associated with the volume found. However, the error is very miniscule. The polypropylene cylinder and acrylic rectangular prism have rounded edges, which throws off measurements. The copper and acrylic prisms also have inaccurate dimensional measurements, since they were intended to be cubes in the first place. Error with the electric balance could be an issue when concerning a smaller decimal place; the balance only measures to the nearest tenth of a gram when other balances could measure smaller. I feel that the balance measurement to the tenths place is the biggest source of error. If the balance measures the mass of the objects just one more decimal place (hundredths), it would have made a big difference in the percent error already. To achieve better results, I would find an electric balance that would measure the mass of an object to the nearest hundredth place instead of the nearest tenth place. This would get me closer to the accepted value for density.
References
http://www.engineeringtoolbox.com/density-solids-d_1265.html
http://lahsphysics.weebly.com/measurement-lab.html
http://lahsphysics.weebly.com/measurement-lab.html